|

B, page 34.—DR. ROBERT JOHNSTON'S SETTLEMENT.
Dr.
Jobnstones Will was dated from London, 30th September, 1639 ; and the
following is the passage endowing the Moffat Grammar School:—"I give and
bequeath unto the Right Honourable Lord Johnstone, One Thousand Poundes
sterling, to bee ymployed in purchasing of landes, for the mayntenance of a
Grammar Schole in Moffett, in Annandale, and doe appoint out of the same
vnto the Mr. ycarely, five hundred Mares, usual money of the Realme of
Scotland, unto the Vsher, and Hypodidusculos, twon hundred mares of like
money yenrely, an vnto an able man to teach the scholiors of the said schole
Arithmetic and Writingo one hundred and ñftye marks of like money of
Scotland, yearlie, and the surplusage of the rent I doe appoint, to be
distribted amongest the poore of the parishe of Moffett aforesaid, and
towardes the reparacon of the said achole, and my mynde is that the said Mr.
Vsher, and Arithmaticians, bee chosen good and sufficient men by the
Provost, Balifl'es, and Ministers of Edinburgh, and also altered by them
upon the inforniacon of the said Lord Johnestoun, his heyers, and the
minister of Moffett, for the tyme being of their insuffielencie and neglect
of the instrucion of the youth there, and others to be elected in the place
of them that bane been caries and negligent. And my will and mynde is that
fiftye pounds starlingo be prescntiie laido unto the said Lord Johnestoun,
towardes the building of the said schole at Moftett, and the foresaid
principall some six monthes after my decease. That the building may be
perfected out of the vse of the said sum before the purchasing of the landos
for the yeerlie maintenance of the said schoic."
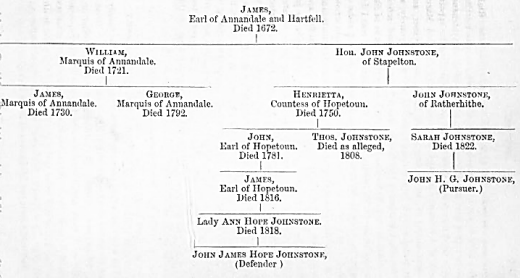
C, page
51.—LINE OF DESCENT OF THE JOHNSTONE FAMILY
The following
is the line of descent of the Johnstone family, as represented in a recent
lawsuit between John Henry Goodinge Johnstone, pursuer; and John James Hope
Johnstone, and Earl of ilopetoun's Trustees, defenders, before the Supreme
Courts, for the title formerly possessed by the representatives of the
family, and which became dormant in 1792, by the death of George, third
Marquis of Annandale, as shown in chapter v., for report of which see
"Dunlop and Bell's eases."
The case was productive of no good, as neither of the claimants had their
hopes or expectations realised, which makes us the more regret with Sir
Walter Scott, who says:—" Every Scotch-man must regret that the name of
Johnstone should have disappeared from the peerage; and hope that some of
the many claimants for the minor honours of the house of Annandale, may make
Out a case to the satisfaction of the House of Lords. The great estates of
the family are still nearly entire, and in worthy hands; they have passed to
a younger branch of the noble house of Hopetoun, one of the claimants of the
elder title."— Vide Note K. to Scott's "Fair Maid of Perth."
D, page 80.—ANALYSIS OF THE MOFFAT SULPHUREOUS WELL
The
following formerly appeared in 1858 In a work, "Moffat: Its Walks and
Wells," and has been procured through the kindness of Messrs. Blackie &
Sons, Publishers:—
The Well, par excellence, is at the
distance of about a mile and a half from the village, and has been generally
described as to situation, &c., in the descriptive sketch of the district.
We would add our testimony to the efficiency of the menus by which the water
is carefully collected at the well for the use of the visitors, and the
orderly and cleanly manner in which it is distributed (by Mrs. Clark) to
those who are In the habit of partaking of it. The walk to the mineral-water
supply is particularly pleasant and picturesque, and from the altitude of
the site of the well, it being about 640 feet above the sea level, the air,
even in the middle of summer, Is never experienced as sultry, at the hour at
which visitors are accustomed to wend their way upwards to the Spa, but is
always, in dry weather, of an agreeable and bracing character.
There are two sources of the water at the well, rising from the rock from
which the water issues, and situated at a few feet from each other. These
are known to the keeper of the well as the upper and lower springs. The
lower one alone is used for drinking purposes. The upper, and to the taste,
and specific gravity, the stronger of the two, is allowed to pass by means
of pipes towards the reservoir, which supplies the mineral baths In the
village. The waste-water of the lower well is allowed to flow in by the same
channel, and adds itself to the contents of the reservoir. From the
well-understood action of sulphureous waters on metals, it would be
advisable that the metal pipes, at present in use for conveying the water
for bath purposes, should be replaced either by pipes made of glass, or,
what would be more economical, and quite as efficient in preserving the
purity and strength of the water, gtua-percha. The tank of the bath
committee, which, we believe, is constructed of cast-iron, ought to be lined
with an inner sheet case of the same material. By such improvements, the
sulphur constituents of the water would sufFer no decomposition on
transference, but would pass on, and be accumulated in the state in which
they exist in the well-water.
On reaching the well, many
circumstances strongly indicate the sulphureous nature of the water. The
water itself has the characteristic odour of such waters, while the metal
stop-cock attached to the pipe which communicates with the rock, and
delivers the supply, is coated with a black shining sulphuret, formed by the
suiphuretted hydrogen disengaged from the water acting upon the metal. The
blackening of the watch-cases, and any silver coins which have been for some
time in the possession of the well-keeper, bears testimony to the presence
of this gas in considerable abundance in the neighbourhood of the well, as
it is evolved from the water when drawn. The small openings in the rock,
from which the water of the upper well issues, are alone visible ; those of
the lower well being built over with a fixed pipe communicator and
stop-cock, to draw off the water at pleasure. The upper and exposed
apertures are incrusted with a yellowish-white substance. This, when dried
and ignited, yields a blue flame, gives off a suffocating odour of burning
sulphur, and leaves a reddish-coloured residue. The composition of this
deposited matter was found, on analysis, to be sulphur and peroxide of iron
(red oxide). In the exposed part of the channel, through which the water
flows from the upper apertures towards the pipes which supply the public
baths, opalescent filaments of sulphur exist in abundance, and also a
considerable quantity of a black deposit. This black substance, on
examination, proved to be the pure protosulphuret of iron. When heated with
hydrochloric acid, it gave off suiphuretted hydrogen gas in abundance. The
acid solution obtained in this experiment contained iron wholly in the state
of protoxide. This was ascertained by Its yielding an abundant blue
precipitate, with red prussiato of potash, but none with the yellow
prussiato of potash. The protosuiphuret of iron is formed by the combination
of the sulphur (of the sulphur compounds In the water), with iron which
issues in a soluble form from several small fissures In the exposed rock;
the protosulphuret of iron so formed, being preserved from the action of the
oxygen of the air by the well water which flows over it. The whitish deposit
formerly alluded to, is, as originally formed, the same substance viz.,
protosulphuret of iron, but by being freely exposed in this form to the air,
it suffers decomposition; Its iron becoming per-oxidized, and its sulphur
set free. In the Iron-conveyance pipes formerly referred to, this formation
of protosuiphuret of iron also takes place, and constitutes the black matter
which is found accumulating in all parts of the piping. As this suiphuret is
completely insoluble in water, it is of no medicinal use in the bath, and
being produced at the expense of much of the all-important sulphur
constituent, its present production is the ground of the recommendation
formerly given for the replacement of the Iron by gutta-percha pipes.
As to the smell and taste of the water, various opinions exist. Dr. Garnett
described it as having a "strong smell resembling bilge-water, or the
scourings of a foul gun," and "like the sulphurcous water of Harrowgate,
though not quite so strong.* The odour is Certainly not the most agreeable,
but this description of it is rather exaggerated. The rinsings of a
gun-barrel do approximate to the odour, but the best analogue, we think, is
a aug/al4, putrescent egg. The taste is that of a mixed saline and
suiphurcous character, so that the water is rather unpleasant to most
palates on first partaking of it. The coldness of the water, however, and
the evolution of minute globules of gaseous matter from it, as well as from
the drinking being fashionable, and consequently a great number of active
examples set before the eyes of the recipient, also the hearty appreciation
of its excellencies expressed by, and the certain hope of expected benefit
so forcibly depicted in, the cheerful countenances of the crowd around him,
soon reconcile the novice to its, on first acquaintance, somewhat repulsive
odour and taste. From our experience of the water, after a week's trial,
both taste and odour, as far as unpalatableness was concerned, had era that
time almost disappeared. It is worthy of remark, that persons once
accustomed to such a water, prefer it, even for constant use, to all other
varieties: pure waters becoming even unpleasant to them. The odour of the
Moffat sulphureous, as of similar waters, is very persistent. It is
detectable soon after partaking, in the expired air on breathing. It passes
off in perspiration, and attaches itself perceptibly to the stockings and
clothes of the water-drinkers, and this, apparently, more or less, according
to the quantity of water the individual partakes of.
The
temperature of the Moffat suiphureous water seems to be quite independent of
that of the external atmosphere; and this during all seasons of the year.
Dr. Garnett states that in the year 1797 he made two observations at
difFerent times on this. In one instance the temperature of the Spa was 50
deg. Fah., while the external air was 54 deg. Fah., and the adjoining brook
48 deg. Fah. On another occasion, when the temperature of the air was 60
deg. Fah, that of the Spa was 49 deg. Fah. Dr. Thomson has not recorded the
temperature at the period when his analyses were instituted. Mr. James Clark
and the writer made the following observations. The method of determining
the temperature was simply to place the bulb and lower end of a thermometer
in a tumbler, and to cause the water, as it issued from the pipe
communicating with the spring, to run continuously into the vessel, and on
the immersed portion of the thermometer for several minutes, the indication
being read off without withdrawing the thermometer from the water

From those
results, and those of Dr. Garnett, the prevailing temperature of 49 J dog.
Fah. may be reasonably considered as having been constant for at least a
period of more than half a century. This constancy of the temperature
furnishes a strong presumption of there existing very great similarity in
the situation and Circumstances under which the water becomes impregnated,
and has bestowed upon It its characteristic properties. The moderate
coldness of the water renders it, as may be conceived, pleasant, at all
events refreshing, during the summer months.
The quantity
of free gas evolved from the water is at no time great. The water Is never
decidedly sparkling. It does assume a certain degree of cloudiness from the
uniform diffusion through it of very minute gaseous globules. These globules
exist more abundantly, and of greater size, on certain occasions than on
others. In short, their comparative abundance may be said to prognosticate
the state of the weather. Mr. Clark, keeper of the well, uses the water as a
weather-glass, and places great confidence in the indications yielded by it.
It Is reported in the locality that when the atmosphere is highly
electrical, as Immediately preceding thunder-storms, these globules are more
than usually almndant.. They are also more abundant, and appear more
rapidly, the higher the temperature of the air to which the water Is exposed
just when taken from the well. A fall of the barometer, indicating a
diminution of atmospheric pressure, and usually a precursor of rain, Is
always accompanied by a greatly increased evolution of gas globules. The
altitude of the situation of the well (040 feet above the ,ea-level) adds to
this sensitiveness of the water to changes in atmospheric pressure. There is
a prevalent error in the district as to this aerated condition of the water.
When the gas globules are larger and more abundant the water is popularly
said to be MqMy charged with its gaseous principle constituent, the
sulphuretted hydrogen. The opposite of this is the proper rendering of the
phenomenon. The increased abundance of these globules is indicative of the
rapid escaping, and consequent loss of the much desired gas. For the same
reasons, the carbonic acid gas in a glass of champagne, or of soda-water, is
much greater in quantity before the condition of effervescence is
established in such liquors by agitation, or by being placed tinder the
receiver of an air-pump, than during the period when the effervescence or
escaping of the gas is going on, or after it. The well-water should,
therefore, be drank as rapidly as possible after being put into the hands of
the visitor, and not walked about with, and thereby shaken and exposed to
the air. The promenade should follow the drinking of each tumbler of water,
it is injudicious to attempt to enjoy both simultaneously.
The statements made as to the temperature and pressure of the air affecting
the retention of the gas, with other obvious reasons of a dietetic kind,
point Out the morning as the best period of the day for obtaining the water
richest in its gaseous sulphur constituent, and that the water is most
valuable for medicinal or therapeutic purposes when the barometer is high.
No dry morning should therefore pass without the invalid remembering to be
at the Spa. On the other hand, on wet mornings, when the well cannot be
conveniently visited, these facts may afford some consolation to the
health-seeker, the water being then less charged in reality, although
apparently more so.
Under all circumstances, the mineral
water must be drunk at the well, in order that the maximum value and effect
of its medicinal character may be obtained. This is over and above the
obvious physical advantages derived from the bracing morning walk We were
told when residing in the village, on reliable authority, that some
visitors, we regret to say of the fair sex, indulged in the highly
objectionable practice of partaking of the water in bed. The water was
conveyed to such soporific drinkers by a messenger sent, to the well for it,
From our own experience, wo frequently saw water being carried from the well
in the direction of the village, no doubt for the use of persons either
unable, and so excusable, or unwilling, and so inexcusable, to do themselves
the benefit of the prescribed, and, we might say, in most instances,
Indispensable morning walk. We were further informed, that those persons who
unwisely partook of the water in the horizontal position consistently also
continued their town practice of partaking of breakfast in the same
situation and circumstances, judiciously, however, allowing an interval of
about two hours to elapse between the water-drinking and the breakfasting,
and afterwards rose when the sun was nearly in the meridian. We do sincerely
hope and believe that such cases are in the most marked manner exceptional,
since so exalted an instance of, we had almost said stupidity, and so gross
a violation of even a common-sense view of the necessary condition of
healthful existence cannot, for the sake of the Individuals themselves, and
for the reputation of the habits of the village, be sufficiently
discountenanced and condemned. These remarks have no reference to those who
are enfeebled from ill health, and cannot undertake the walk, perhaps not
even the well-omnibus drive, but apply to, we trust, the very few, who,
while quite able to do so, still from indolence remain at home, and drink
their potations there. Much of our urban usage must be left in the city it
we would recruit health in rural Motrat, or reap advantage from its
mineral-waters.
The specific gravity of the sulphureous
water of the well, as indicated by the use of a superior hydrometer, was
fully 1.001. By the use of a 500-grain sp. gr. bottle, the specific gravity
of the water was found to be .00124, and in another trial with a 1000-grain
sp. gr. bottle, 1.00122. The mean specific gravity of the two last and most
delicate experiments, would therefore indicate 10023 as the specific gravity
of this cater. Due observance was made of temperature and atmospheric
pressure in these experiments. The specific gravity of the water of the
upper well, which is totally employed for bath purposes, was slightly above
that stated.
The qualitative chemical analysis of the
water was conducted at the well, by means of suitable apparatus and
re-agents.
The ingredients found present in the
sulphureous water were sulphur, lu the form of sulpliuretted hydrogen gas,
and also as a sulphuret (most probably of sodium), also, chlorine, lime,
magnesia; and, subsequently, there were found in the quantitative analysis,
soda, traces of earthy carbonates, organic matter, and silicic acid. The
substances judged absent, or not present in detectable quantity, were
sulphuric acid, potash, iron in any of its states of combination, carbonic
acid gas, and nitrogen or any other permanent gas. Excepting the traces of
lime and magnesia existing as carbonates, and the soda present in the water
in the form of silicate of soda, the lime, magnesia, and soda were found to
exist as calcium, magnesium, and sodium, in combination with chlorine, and a
small proportion of; most probably, the sodium with sulphur.
It may be remarked that, while testing the well water, most of the tests
were applied to the water of the adjoining burn, and also to that of the
spring situated on the edge of the footpath, between the stream and
esplanade, and leading to the verandah at the well. The burn-water
indicated, in the ucconcentrated form, only traces of chlorine. Chemically,
it is a very soft and pure water, but is coloured considerably from
dissolved organic matters derived from moss. The water from the spring
indicated traces of chlorine, slight traces sf sulphuric acid, and very
slight traces of lime. It is a very excellent and safe drinking water; and,
as a spring-water, it is unusually soft.. Neither the burn nor spring-water
contained the slightest trace of sulphureous impregnation.
By a series of experiments the respective quantities of the soluble salts,
earthy carbonates, and of siliceous and organic matters, composing 50 grains
of the solid residue on the evaporation of the sulphureous water, were
determined; and the results, when calculated to represent their
proportionate amounts in the quantity of residue from the imperial gallon,
were as follow:-

The combined sUicic acid exists in the original water as silicate of soda.
The 206 grains of silicic acid unite with 140 grains of soda, and are thus
equivalent to 346 grains of silicate of soda.
The 226
grains of earthy carbonates were found to be:-

The amount of
chloride of sodium (common salt) thus indicated was 6072 grains per imperial
gallon.
The most important determination in the Inquiry
was the quantity of sulphuretted hydrogen gas present In the Moffat
sulphureous water. The best known processes were instituted to determine
this, and the results of five different experiments obtained. The mean of
all the experiments was 678 grain of sulphur, equal to 720 grain by weight,
and 182 cubic inches by volume of sulphuretted hydrogen gas.
A portion of the well-water, after being heated to ebullition, was found
still to contain sulphur in solution, indicative of the presence of soluble
saline sulpirurets in the original water. This showed that the sulphur does
not all exist as sulphuretted hydrogen in this water, as has been generally
supposed, rind represented in previous analyses. It also accounted for the
slightly alkaline reaction of the aqueous solution of the residual matter
left on the evaporation of the original water. The total snlphur in the
original water was found to be, per gallon, 742 grain, from which, if we
take l.622 grain for combined sulphur, we shall have left, as sulphur
existing in the form of suiphuretted h)drogen gas, .120 grain, which is
equivalent to -128 grain by weight, and 353 cubic inch by volume of this
gas. From this it will appear, that about one-sixth only of Me sulphur in
the well-water exists as a gaseous compound, the other flre-si.rths existirg
an alkaline su-Iphuret. The former is alone expelled or decomposed by heat.
The latter remains in solution oven after the water has been in a state of
ebullition.
From the foregoing results, one imperial
gallon of the Moffat sulphureous water contains, in the form of salts
soluble in water, after the evaporation of the original water to dryness,
the following ingredients, in the proportions slated; the portion of the
residue found thus soluble, being, as before recorded, 8227 grains:--

The bases of
the saline compounds, formed by the combination of the above constituents,
must exist as chlorides, since no other radical or acid is present to unite
with them; excepting the sodium existing as the partner in the soluble
alkaline sulphur salt, and the soda, which is most probably the base, united
with the silieie acid, existing as silicate. The states of combination in
which the ingredients exist in the original water are thus represented on
calculation:-

cous water will, no doubt, simplify the problem as to its therapeutic
properties in the view of the medical man. its composition in this respect
is very similar to the waters of the same class at Aix-la-Chapelle and
Harrowgate. The proportion of sulphur present, equivalent to 2,168 cubic
inches of sulphuretted hydrogen gas, indicates that the Mofrat water is not
one of the strongest of su1phureous waters. The Harrowgate water in England
(the old well) contains a much higher quantity of this gas in proportion to
the other constituents. Enghicn, in France, is more like MolFat in this
particular. The state of dilution in which the sulphur compounds exist in
the Moffat water, should place its internal use completely under the safe
control of the invalid.
The peculiarity, now for the first
time recorded, that five-sixths of the sulphur in the MotFat water exists in
the form of an alkaline sulphuret, and remains in solution after boiling, is
most advantageous and important. The first advantage resulting from this, is
in the external use of the water. The alkaline sulphuret is analogous in its
action on obstinate cutaneous or skin diseases, as lepra and scabies, to the
sulphuretted hydrogen gas; but it is not, like that compound, dissipated or
decomposed on the application of heat to the water. Warm baths of the
Mofl'at water may, therefore, be indulged in, without more than a very
partial loss of its sulphureous impregnation. This anticipation was
confirmed by experiment. The temperature at which the water is so employed
is generally 98 deg. Fab. There are occasionally exceptional cases, but
these are few in number. On submitting a measured quantity of the water to
this temperature for a space of time equal to the Customary bathing period,
and then estimating, by a process previously detailed, the sulphur remaining
in solution, less than one-sixth of this constituent had disappeared,
showing, that besides the whole of the combined sulphur, that even traces of
the gas remained. In heating the water for bath purposes, contact with metal
should be avoided, and wooden bathing vessels should be used. Another most
important advantage arising from the presence of the fixed alkaline
snlphnret, bears on the transmission of water to invalids residing at a
distance from the well. It is sent to such In Glasgow, Edinburgh, Carlisle,
&c. The former has as yet been the principal emporium for It. Were the
sulphur in the water existing solely as sulphuretted hydrogen, this gas
would soon be dissipated, and the water, as regards this Ingredient,
rendered valueless. The agitation consequent on transference would aid this.
The fixed state of the combined sulphur, however, prevents its escape, so
that although part or all of the gaseous sulphuret should be lost, the water
cannot he said to have lost more than one-sixth of its leading therapeutic
ingredient. The full medicinal adrastage, of course, only obtained by those
who veil The well; but the fact now stated is interesting and consolatory to
those, however distant, who cannot, from circumtunccs, have this privilege.
The retention of even portions of the gaseous suiphuret is insured for short
distances especially with railway communication, by careful scaling of the
jars in which the water Is transmitted.
It is not
advisable that the water should be drank after being kept for any great
length of time. The changes In a jar of the water during three months were
carefully observed by the author In his laboratory. The vessel was not
tightly corked. The water became slightly opalescent, and of a peculiarly
offensive odour. After two months this disagreeable odour passed oft; and a
decided vinegar odour was exhaled, with effervescence on agitation, like
that from a very sour fermented liquor; the water at same time being highly
acid to test-papers. In three months this had disappeared; the water was
odourless, and on the application of the usual tests, not a trace of sulphur
in any form, either as sulphuric acid, hyposulphurous acid, sulphuretted
hydrogen, or as an alkaline sulphuret, was detectable. These remarks point
out to the bath committee in the village, as well as to others collecting
similar water for bathing purposes, the necessity of not permitting such
waters to remain exposed to the air for months in tanks, although this may
be found very convenient for accumulation.
Many opinions have been entertained as to the origin of
the sulphureous impregnation of the MofFat well-water. Dr. Garnett
considered it as most probably derived from a bog in the vicinity. This
supposed source has now disappeared, and yet the impregnation continues.
Portions of rock from the strata immediately adjoining the well, on being
examined chemically, were found to contain sulphur in considerable
quantity, in the form of iron pyrites or bisulphurot of iron. The rock
from which the spring has its issue was found not to be free from this
ingredient. This sulphur compound is present in such quantity in some
masses of the slaty rock, as exposed in the bed of the stream above the
site of the well, as to render the crystals of it distinctly visible to
the unassisted eye, and even layers of it, from one-eighth to one-half of
an inch in thickness, traverse these rocks. This sulphuret may indeed be
said to be a prevailing constituent of most of the minerals of the
district, and appears to be the prime source of the sulphureous
impregnation it the mineral water. The situation of the well, lying as it
does at the foot of an extensive series of hills, characterized here and
there by the presence of the metallic sulphuret named, seems to place this
beyond doubt. The constituents of some mineral waters, as chloride of
sodium, carbonate of lime, &c., may be referred to lixivation and solution
merely, the rain-water dissolving such already formed constituents during
its percolation through the mineral matter which contains them. Other
ingredients, however, are the result of chemical decomposition, as well as
subsequent solution. Sulphurettod hydrogen gas and alkaline sulphurets
belong to this latter variety. They are produced by the action of water on
some metallic sulphuret, and most usually that very sulphuret which is so
abundant in the vicinity of the Moirat well. The presence of organic
matter favours, if it is not indispensable to this formation. The oxygen
of the water combines with the metal of the metallic sulphuret, while its
hydrogen unites with the sulphur, forming sulphuretted hydrogen. The
conditions for this chemical action all exist in the case of the well
water. The source of the alkaline sulphuret in the water Is most probably
different. This constituent is most likely referable to the action of the
organic matter, which is present in considerable proportion in the water,
and is of vegetable origin, excepting a few animalcules, on alkaline
sulphates. The oxygen of the sulphate, say sulphate of soda, is removed by
the carbon of the decomposing organic matter, and sulphuret of sodium
results. It may be stated, in objection to this view, that the Moffat
water contains no such sulphates. This is certainly true of the water as
it now issues at the spring. Dr. Thomson records the presence of such
constituents in great quantity at the period when his analysis was
conducted.
In June, 1834,I visited the Sulphureous Well,
and conducted a further experiment, so as to indicate any change since last
season in the sulphureous impregnation of the water. The arsenical solution
was used. In this result, 323 cubic inches of the water were employed. There
existed 165 grains of the tersulphuret of arsenic. This amount, on
calculation, coincides in an exceedingly near, and to me gratifying manner,
with the results of the similar experiment conducted in August last, which
yielded 146 grains of the arsenical sulphuret, from 300 cubic inches of the
sulphureous water. This recent experiment would indicate constancy in the
amount of the sulphureous impregnation in this water from August last till
now, and in this view the result possesses great interest. |
